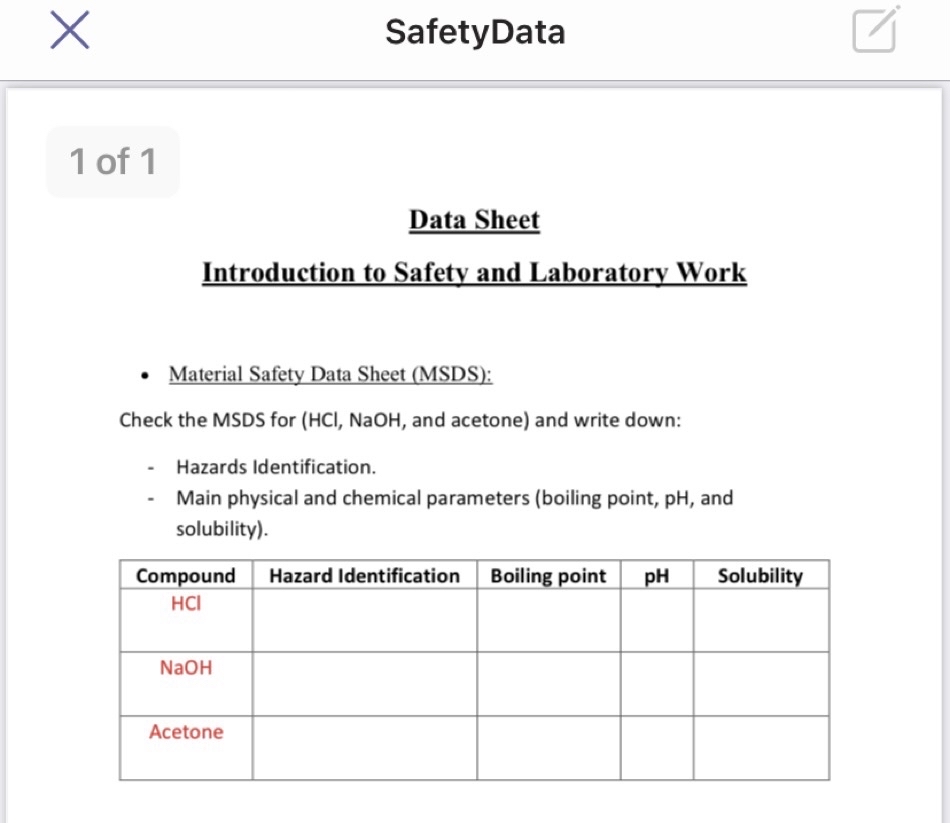
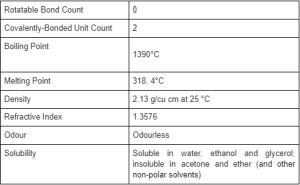
3. The systems acetone–sodium hydroxide–water and acetone–potassium hydroxide–water at 0° - Journal of the Chemical Society (Resumed) (RSC Publishing)

Solubility of Electrolytes in Organic Solvents: Solvent-Specific Effects and Ion-Specific Effects | Journal of Chemical & Engineering Data
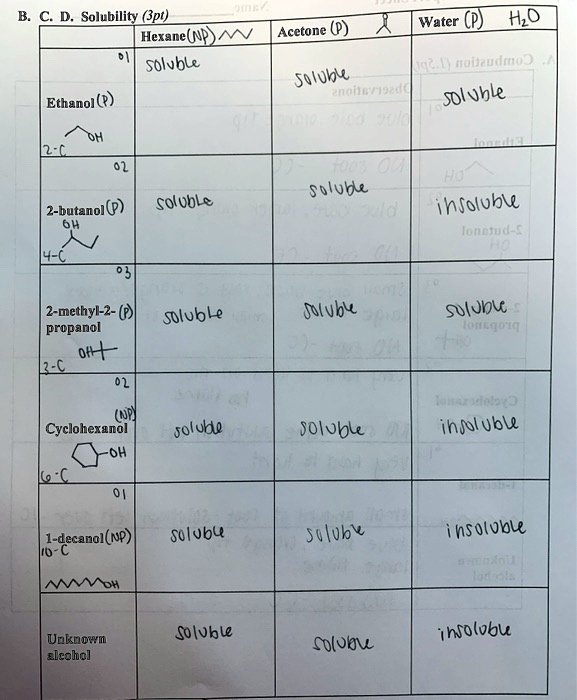
SOLVED: Text: Solubility (pt) Hexane (Not Soluble) Water (Soluble) Acetone ( Soluble) Ethanol (Soluble) 2-butanol (Soluble) 2-methyl-2-propanol (Soluble) Cyclohexanol (Soluble) 1-decanol (Not Soluble) Unknown alcohol (Soluble)
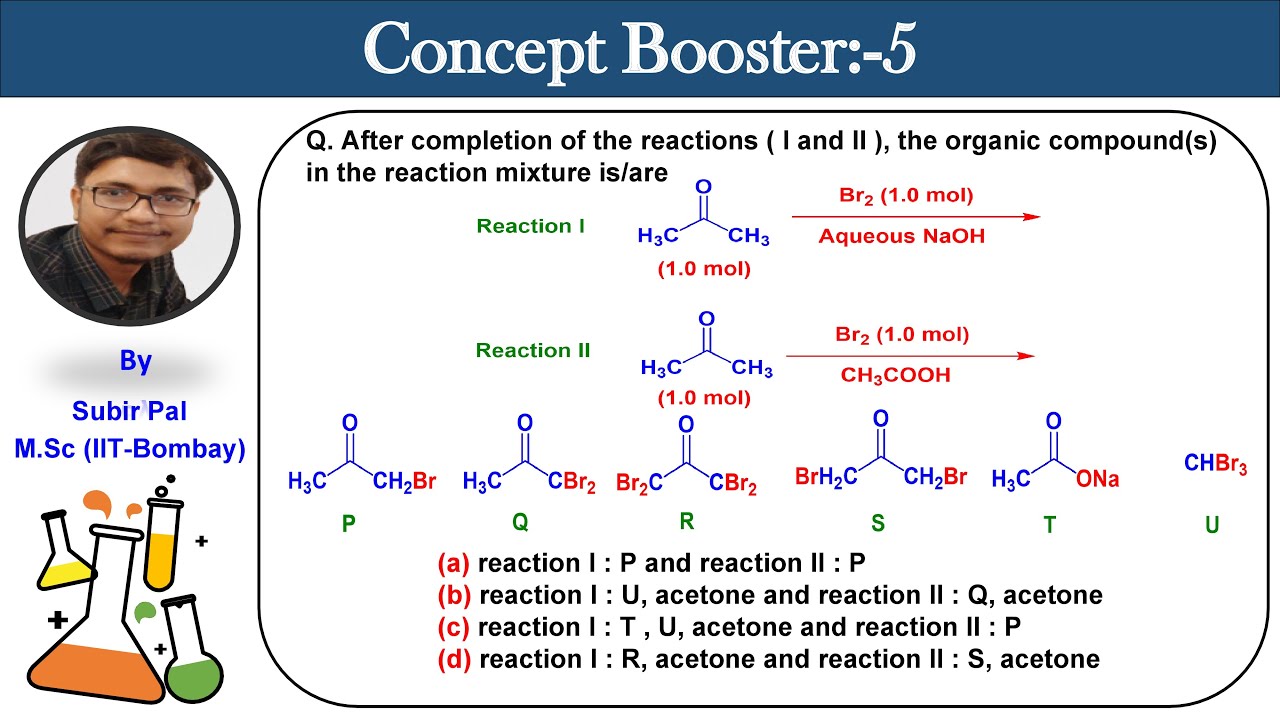
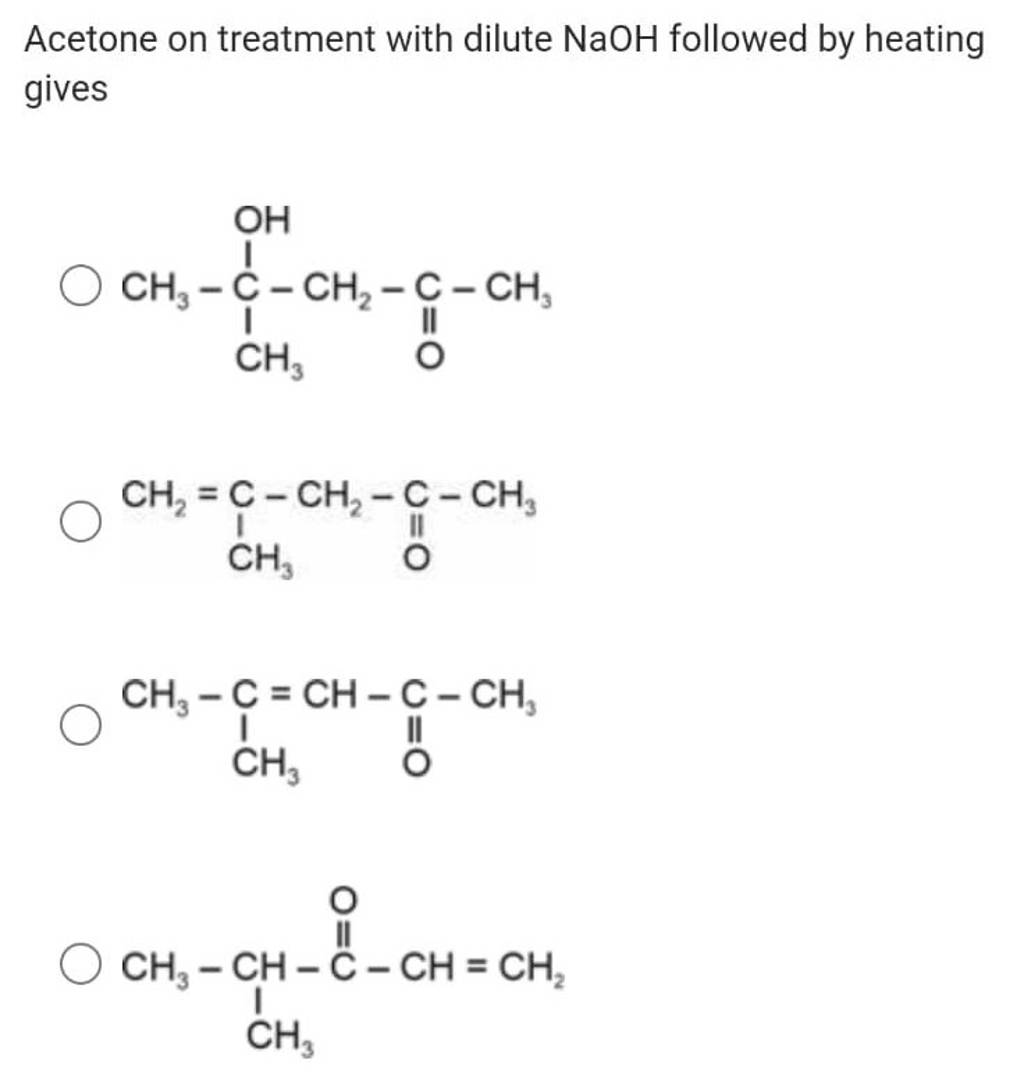
Bromination of Acetone in Aqueous NaOH and CH3COOH || Concept Booster-5 || CSIR-NET || GATE - YouTube
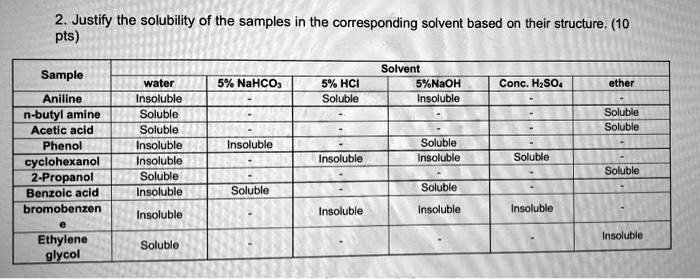
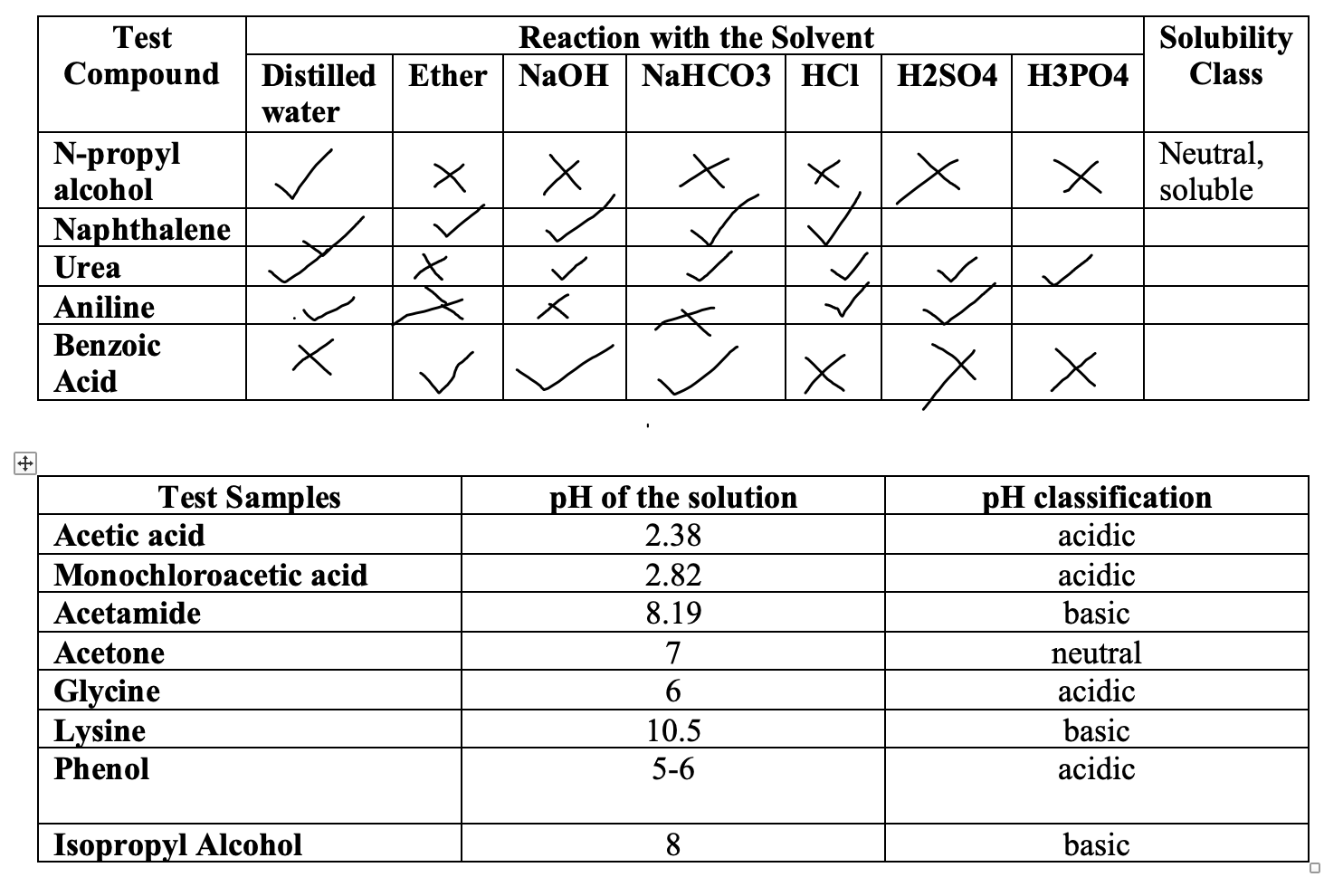
SOLVED: 2. Justify the solubility of the samples in the corresponding solvent based on their structure. (10 pts) Sample Solvent 5 NaOH Insolublo wator 5% NaHCOs 5"k HCI Soluble Conc. HzSO4 Aniline

Effects of NaOH and Ethanol on the Solubility of Disodium Decanedioate in Water | Journal of Chemical & Engineering Data